Highlight
Basic models for proof of concept; Advanced models for translatability into the clinic
Significantly shorter induction period, 12-16 weeks before compound dosing
Clinically relevant models closely mimics human NASH disease condition: dysmetabolic syndromes,
inflammation, hepatosteatosis & fibrosis
Higher and more robust fibrosis score than the current diet-induced NASH mouse & hamster models
Pharmacologic responses of reference compounds like OCA, Semaglutide & GTF505 match responses of
clinical trials
A New NASH Model Induced with Modified HFD Plus Fructose
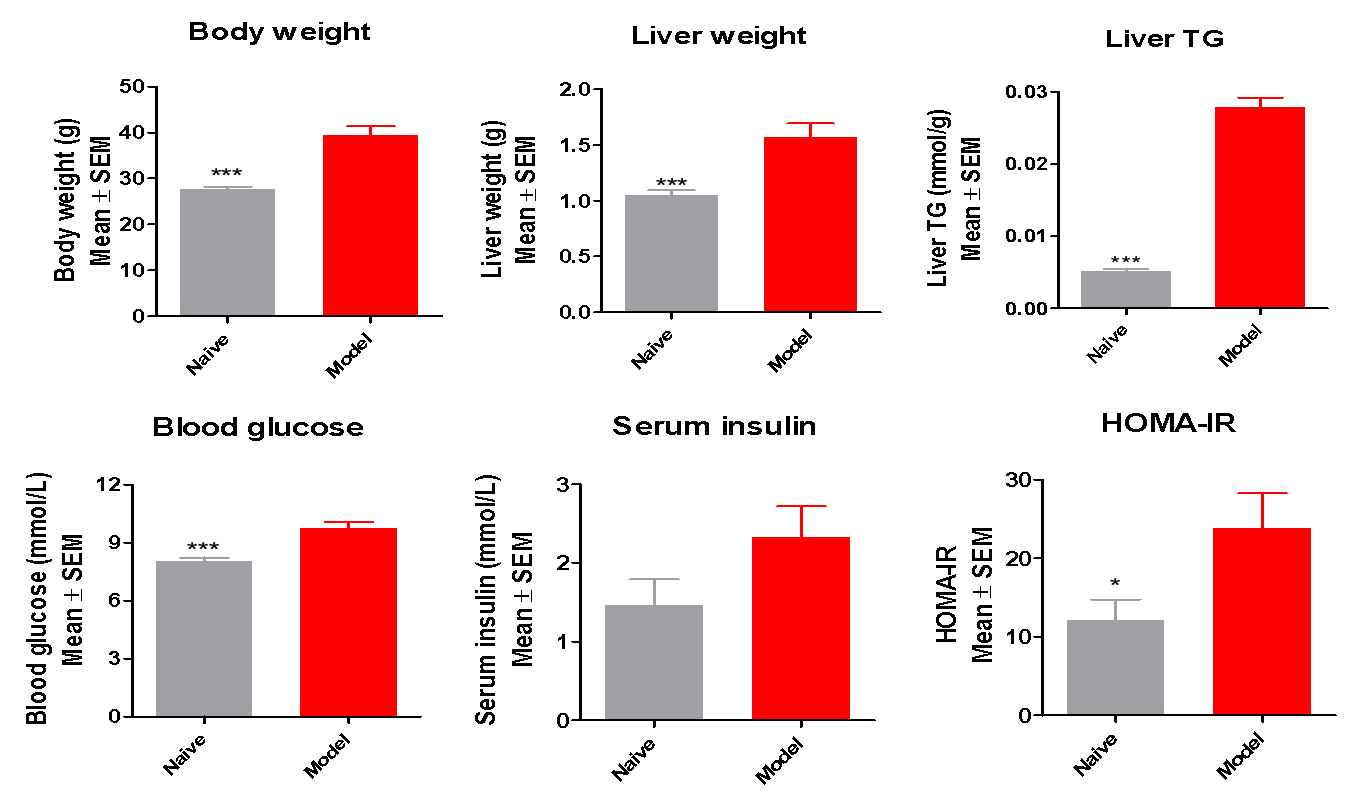
The body weight, liver weight and triglycerides significantly increased in modified HFD induced mice
Glucose and insulin levels also increased in NASH mice
Induction period: 12 weeks
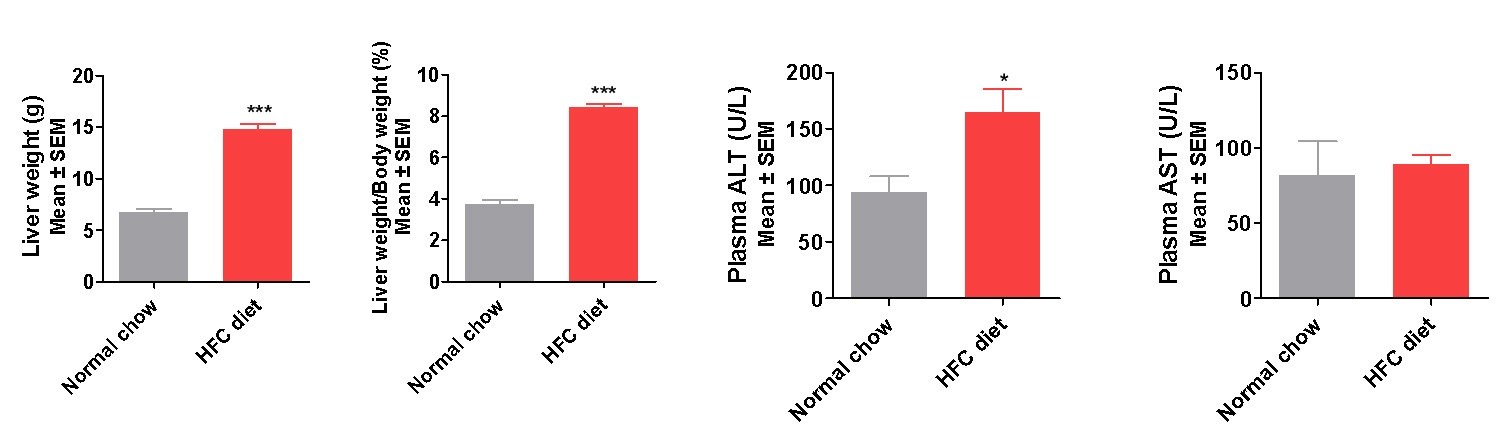
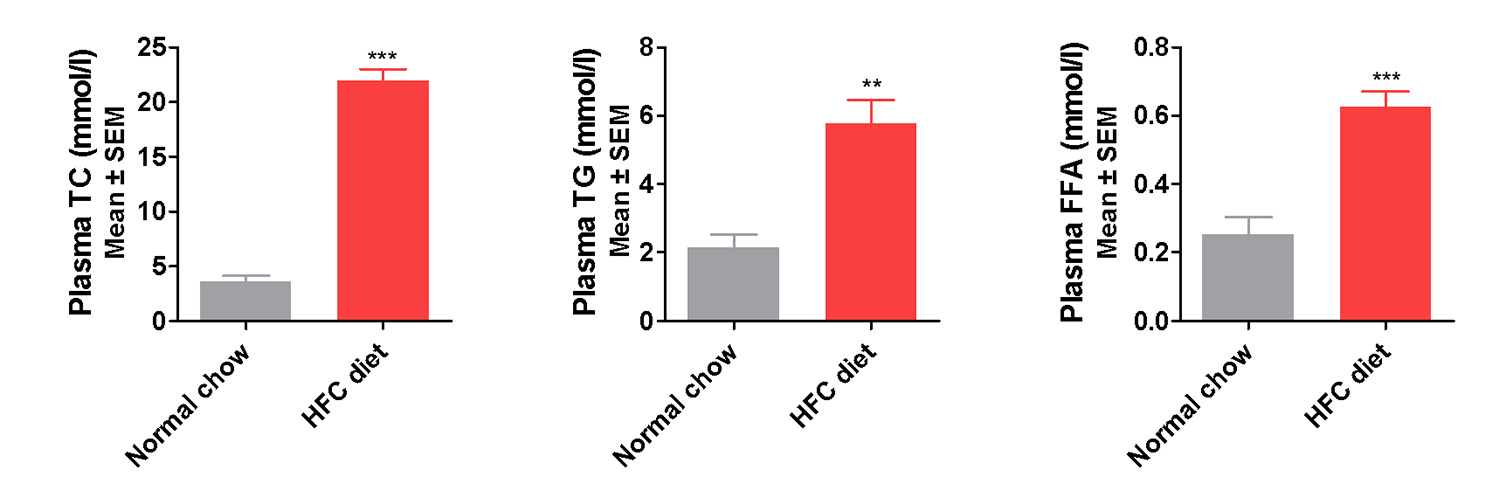
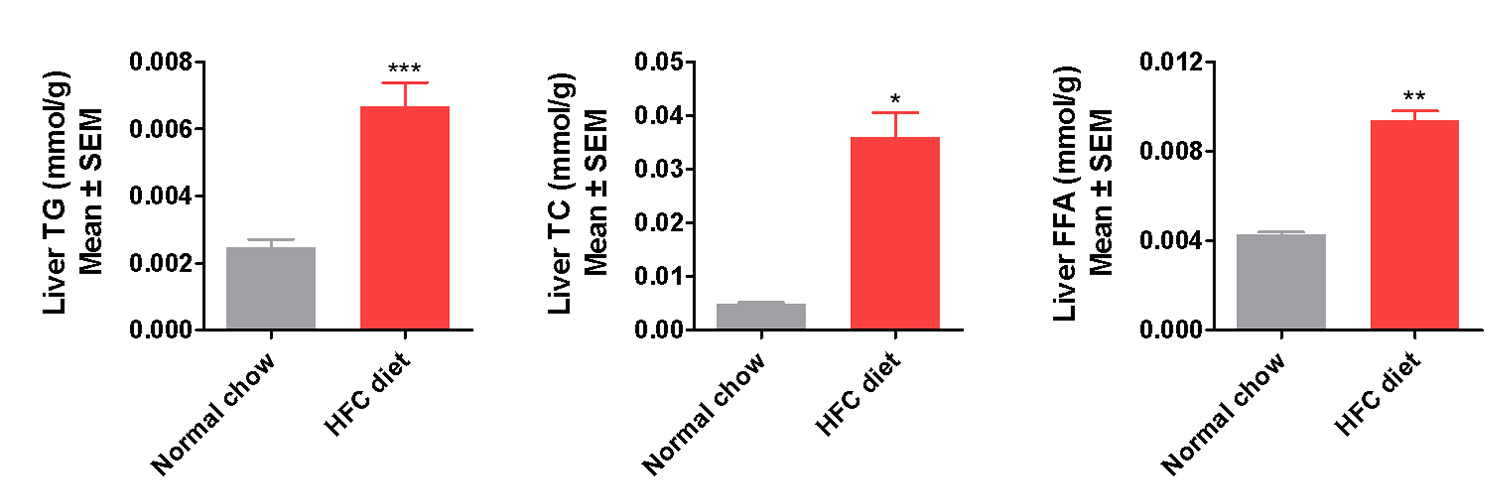
Liver Lesion and Dyslipidemia Manifested in the New NASH Model
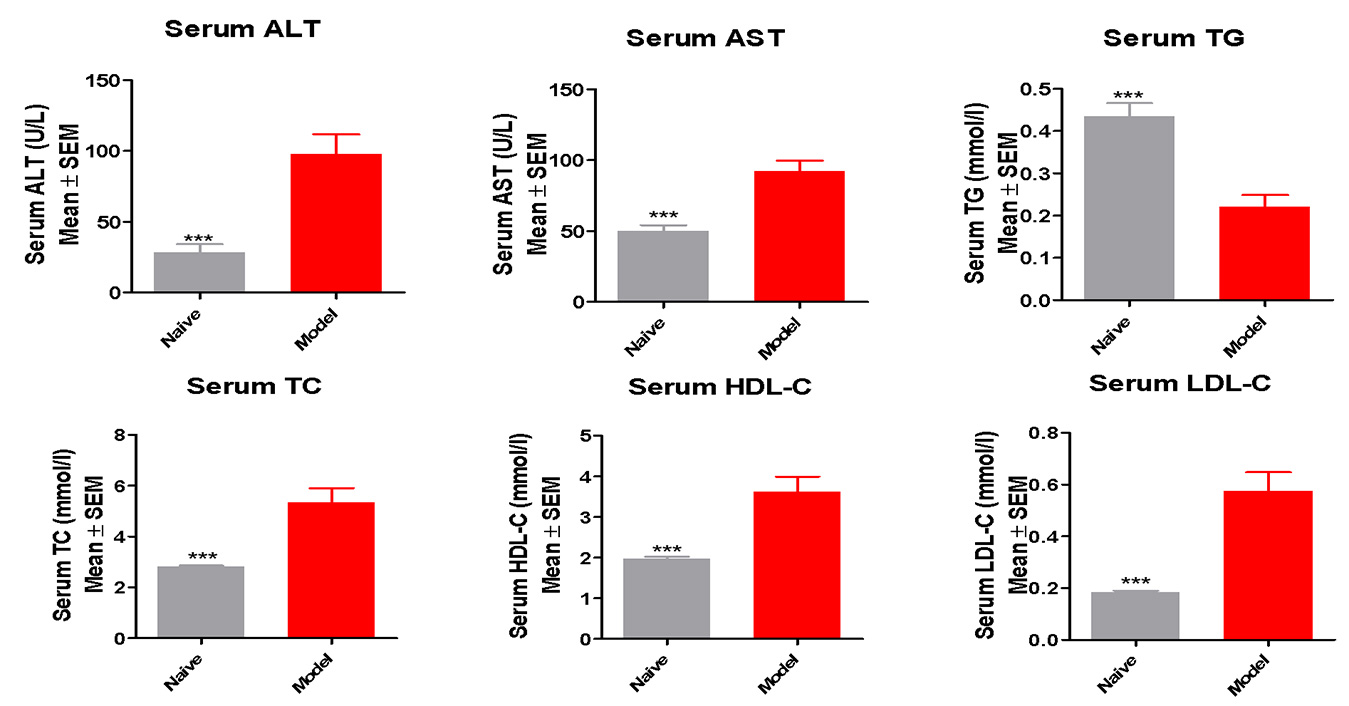
Elevated liver Enzymes (ALT, AST) and serum lipids (TG, TC) indicate liver lesion and dyslipidemia present
in the modified HFD induced mice
Induction period: 12 weeks
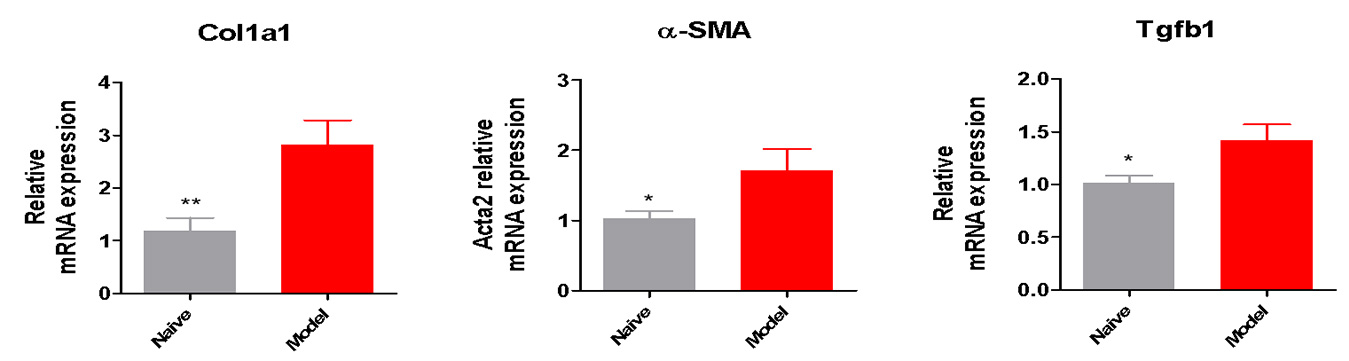
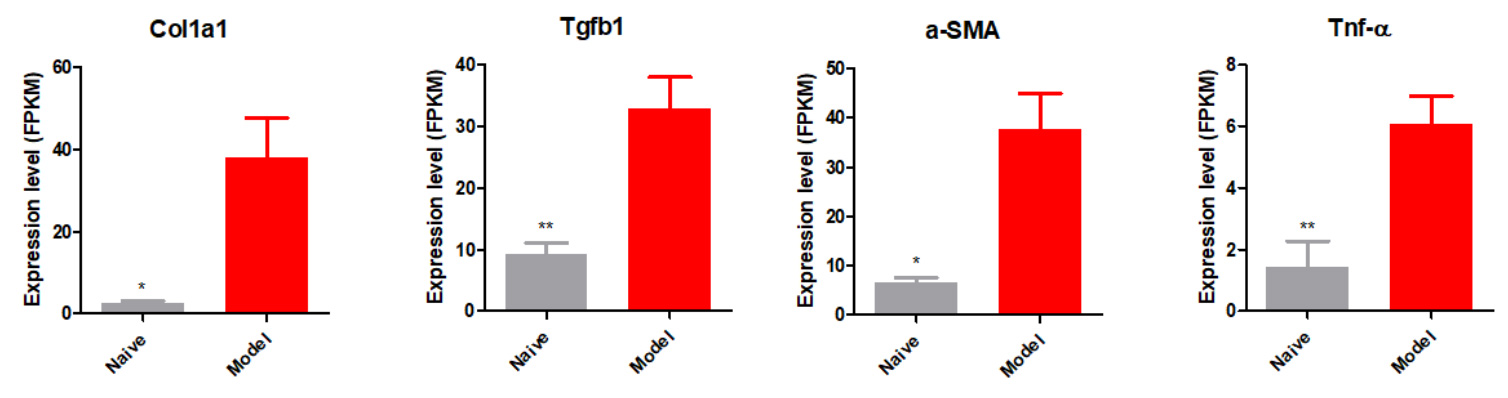
Fibrosis Biomarkers Elevated in the New NASH Model
Relative mRNA expression of fibrosis related genes increased in the modified HFD induced mice
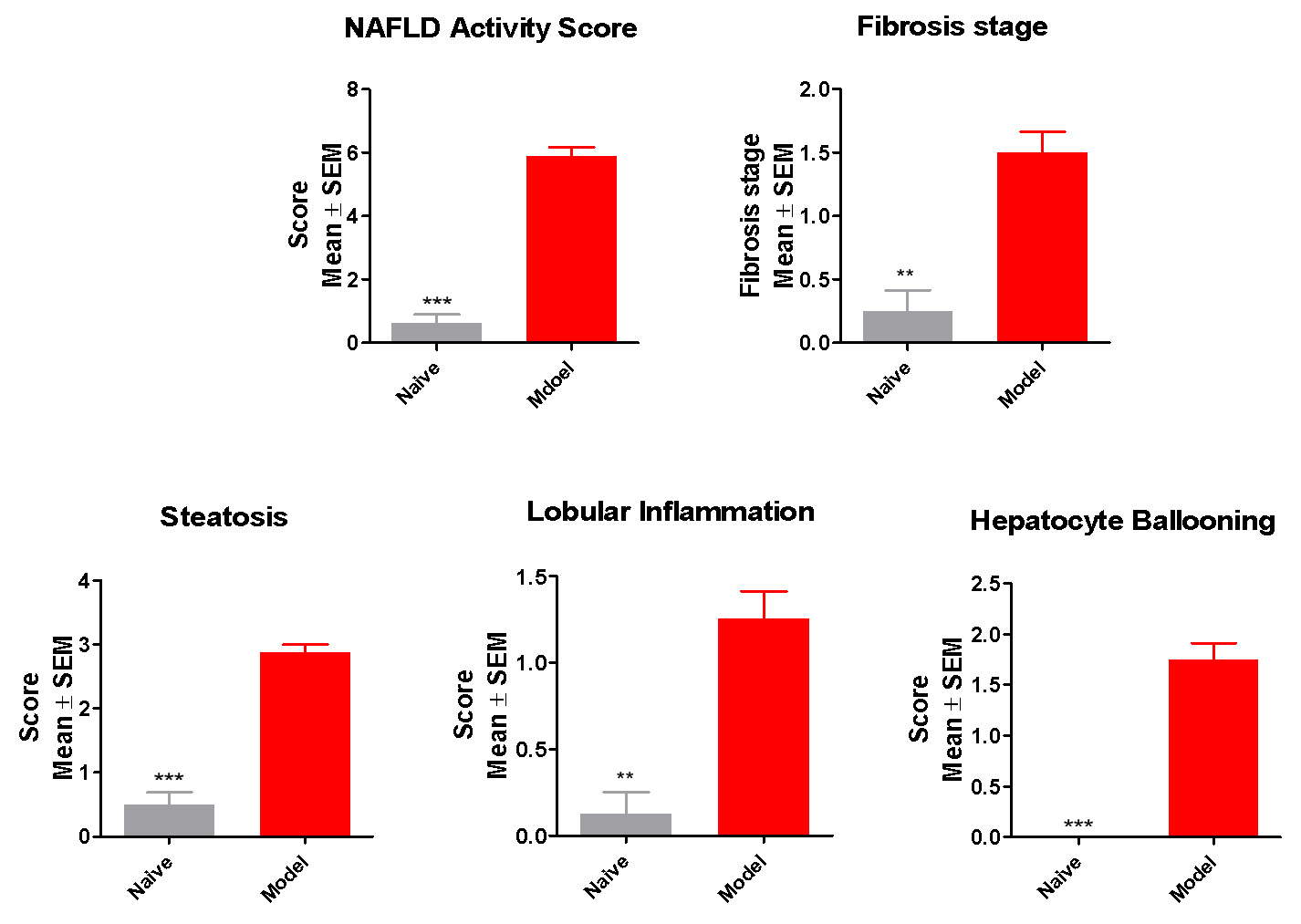
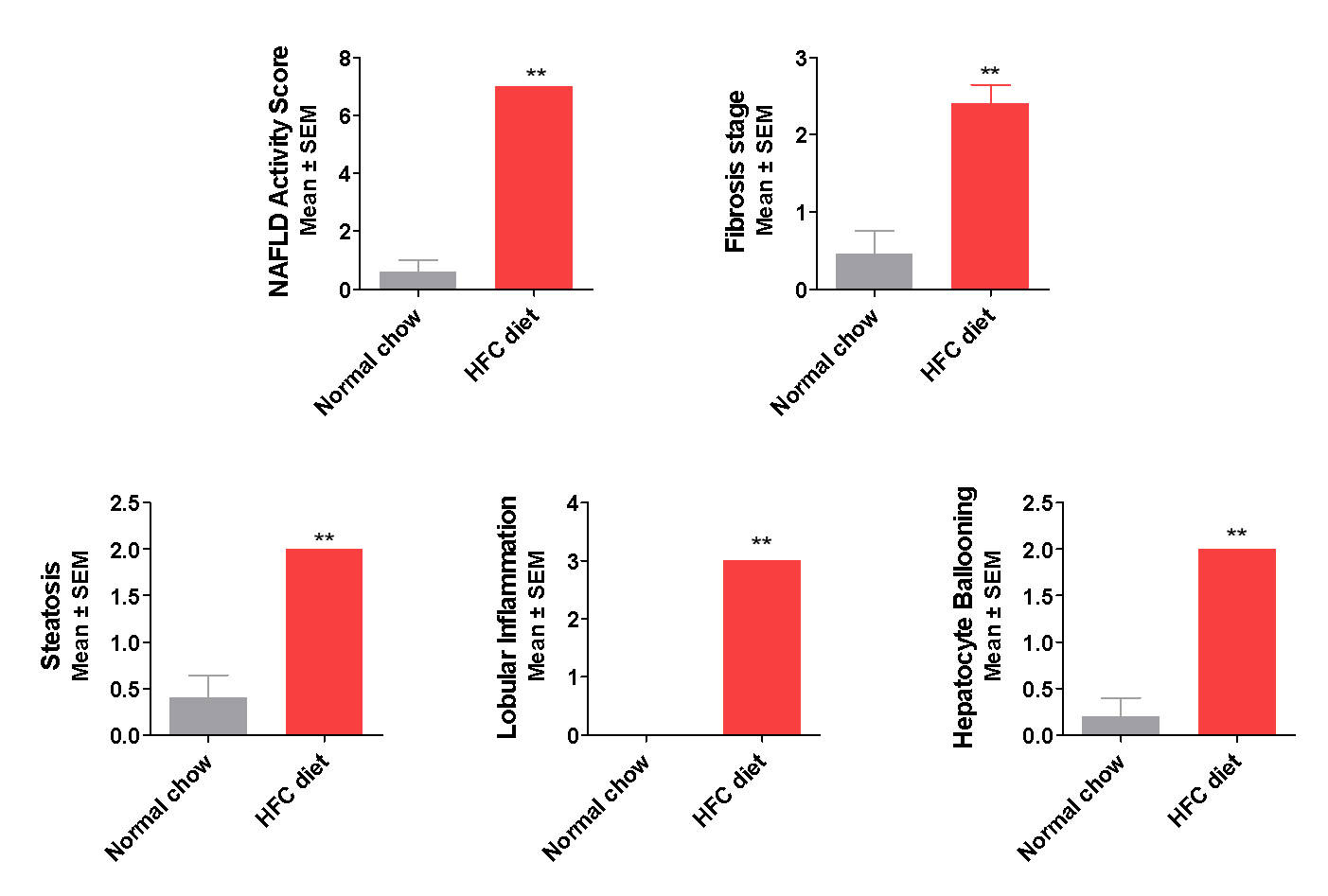
Robust NAFLD Activity & Fibrosis Developed in the New NASH Model
The new NASH model exhibits hallmarks of NASH
Basic models for proof of concept; Advanced models for translatability into the clinic
Insulin
Glucose
Cholesterol
Triglyceride
FFA
Histopathology: increased steatosis, ballooning, inflammation, Mallory body and Fibrosis
The phenotype is consistent with human metabolic syndrome/NASH-Fibrosis
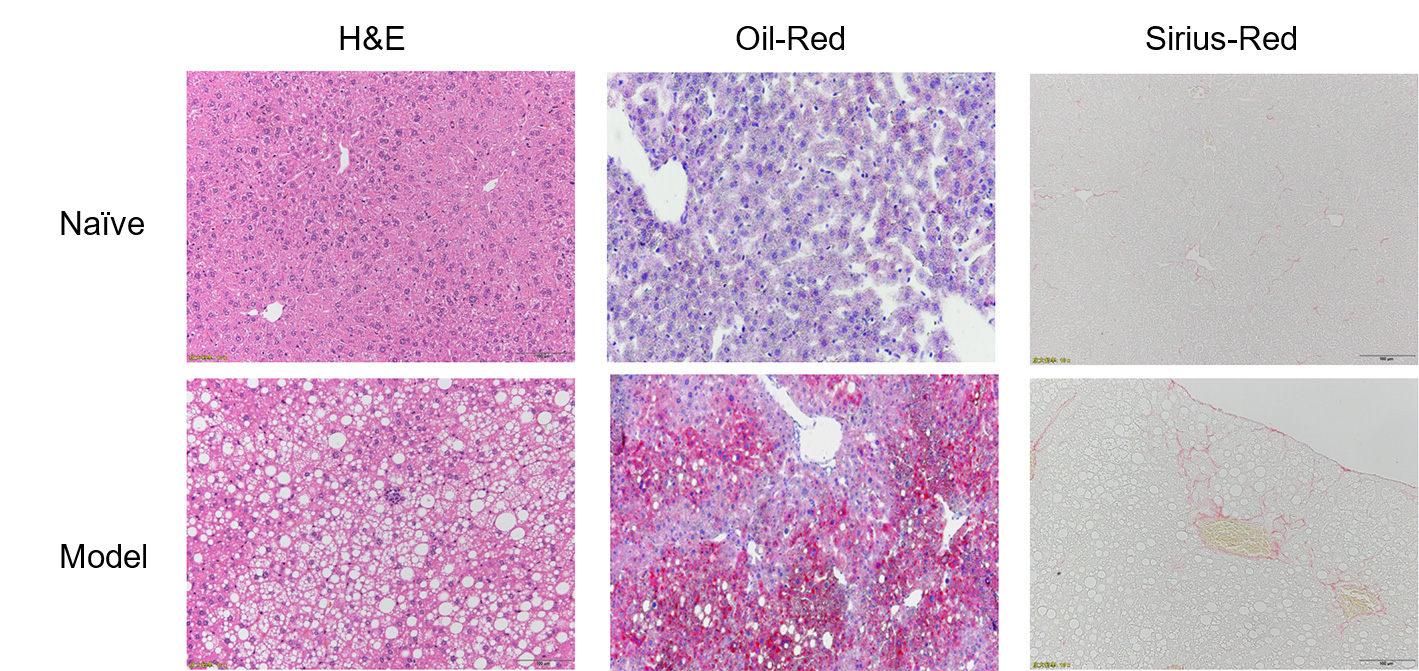
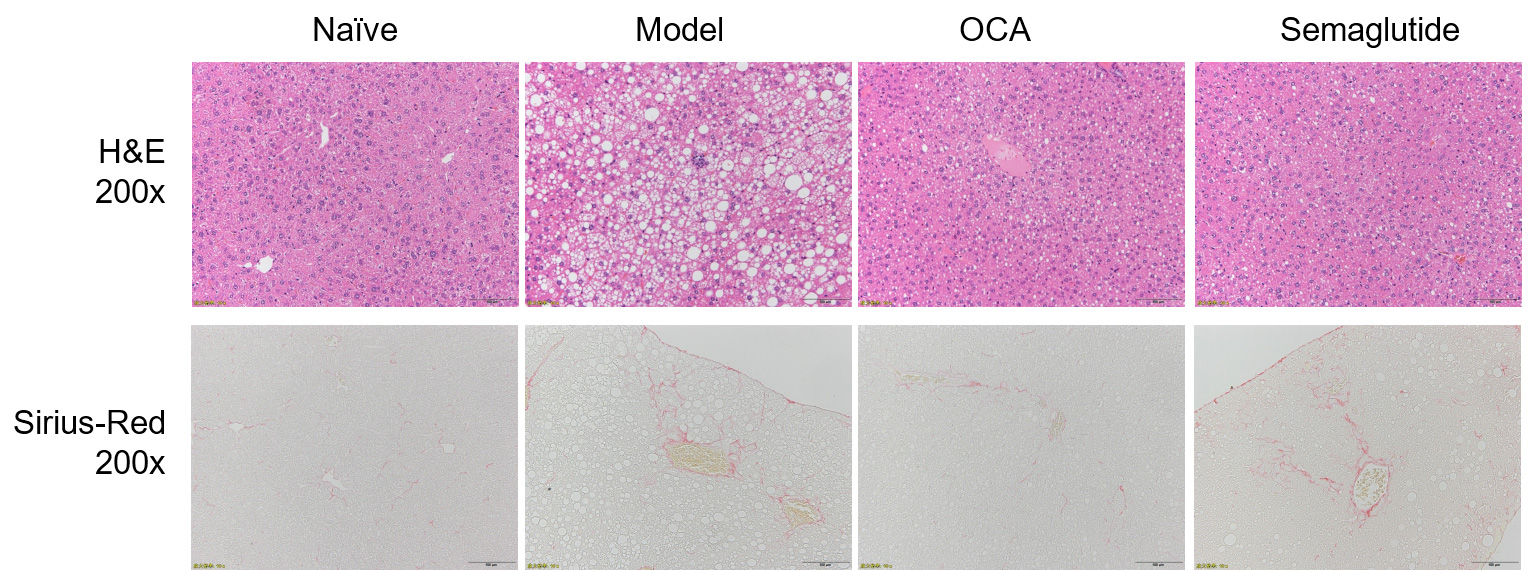
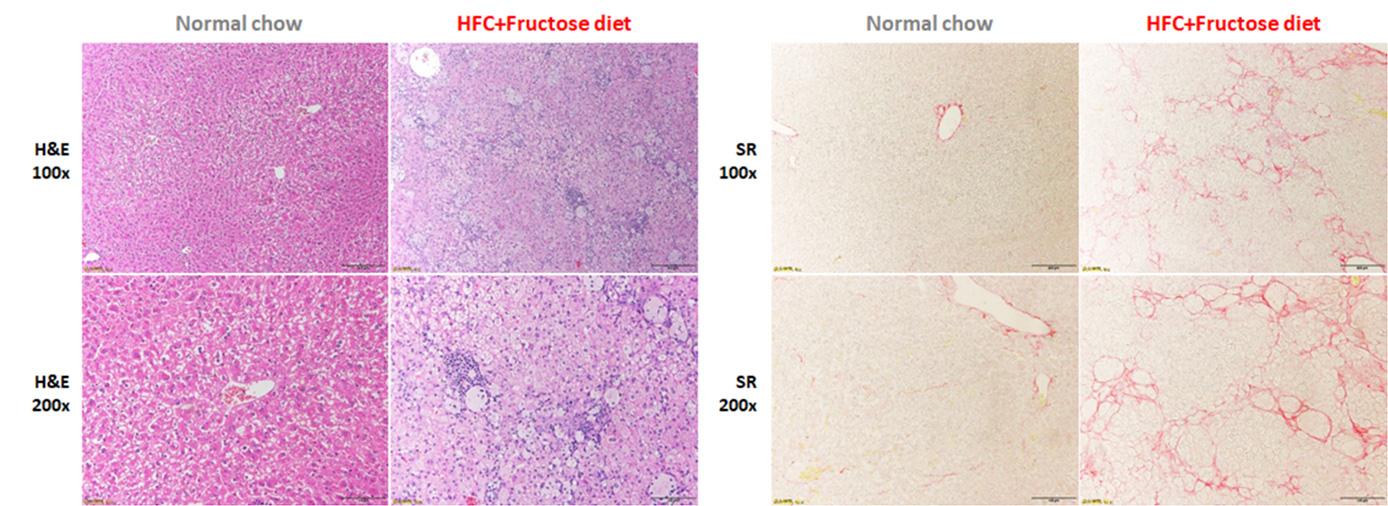
H&E and Oil-Red staining showed steatosis, inflammation and ballooning all developed in the modified HFD
induced mice. Sirius-Red stains confirmed presence of fibrosis
Validation of the New NASH Model with Reference Drugs
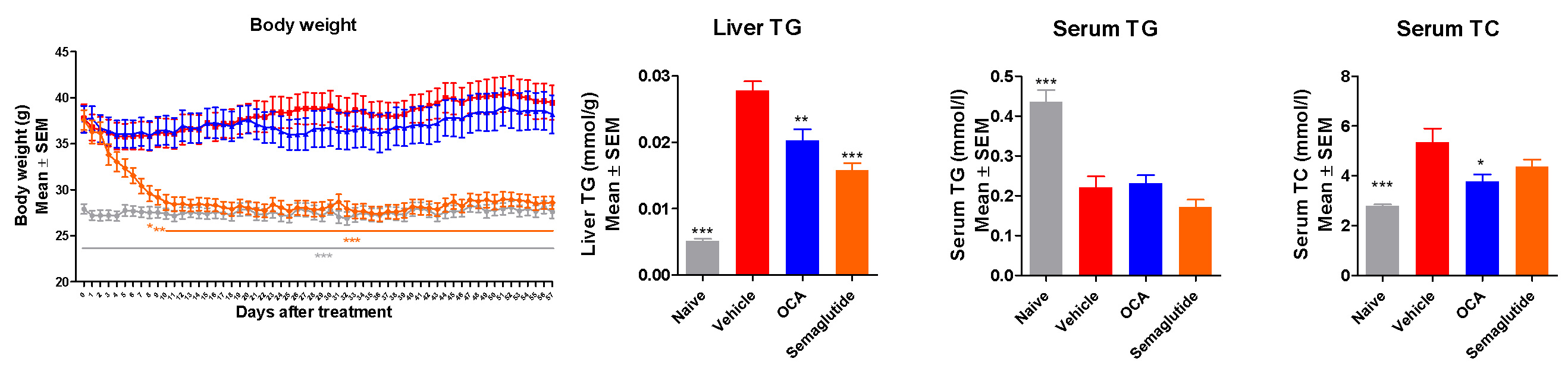
Effects of OCA and Semaglutide on Liver and Serum Levels of TG, TC
OCA: PO, QD, 30mg/kg
Semaglutide: SC, QD, 0.05mg/kg
HFD induction for 12 weeks, mHFD and treatment
for 8 weeks
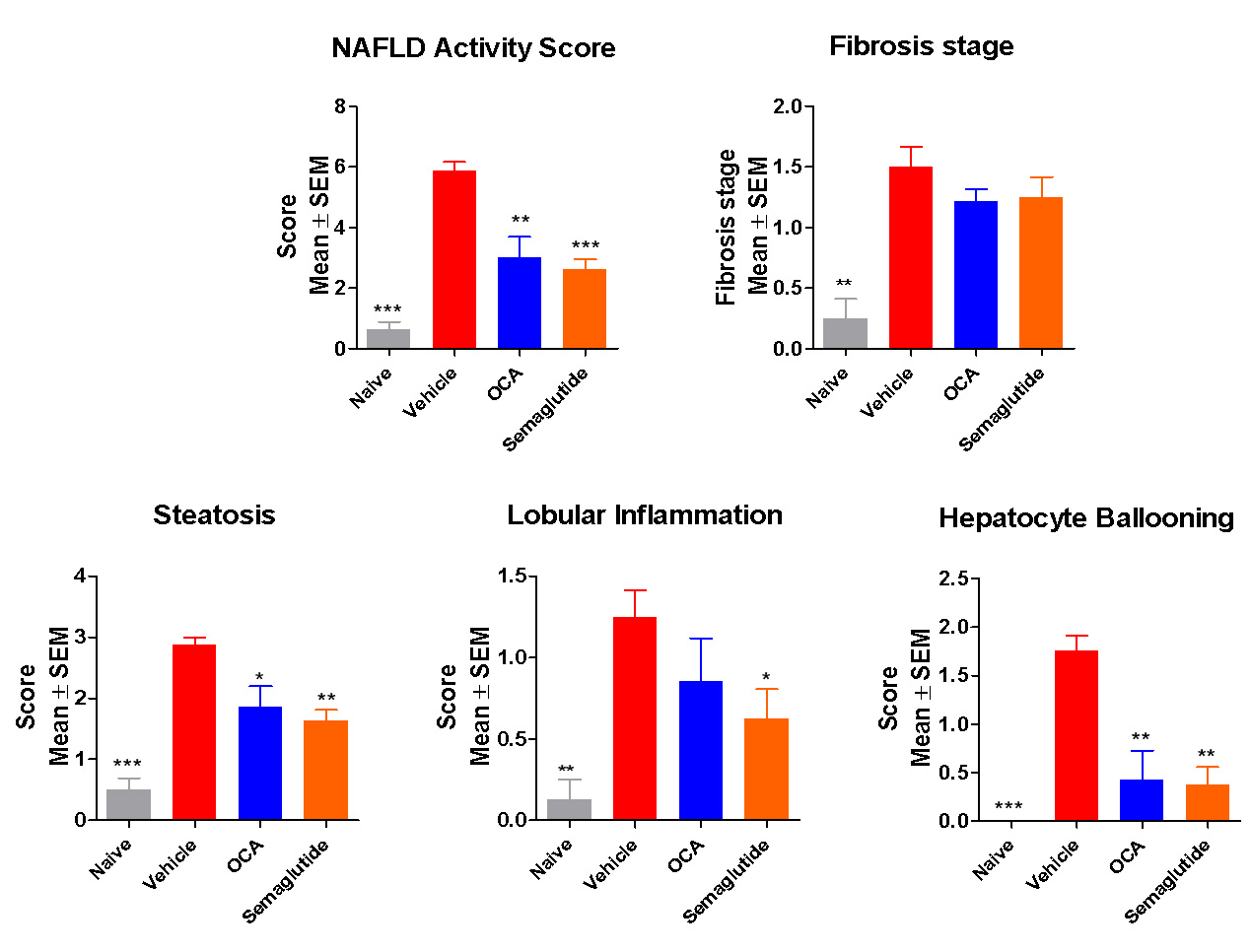
Both OCA and Semaglutide showed significant effects after 8 weeks treatment:
Reduced liver enzymes
Reduced Cholesterol
Lowered BW
Reduced Liver TG
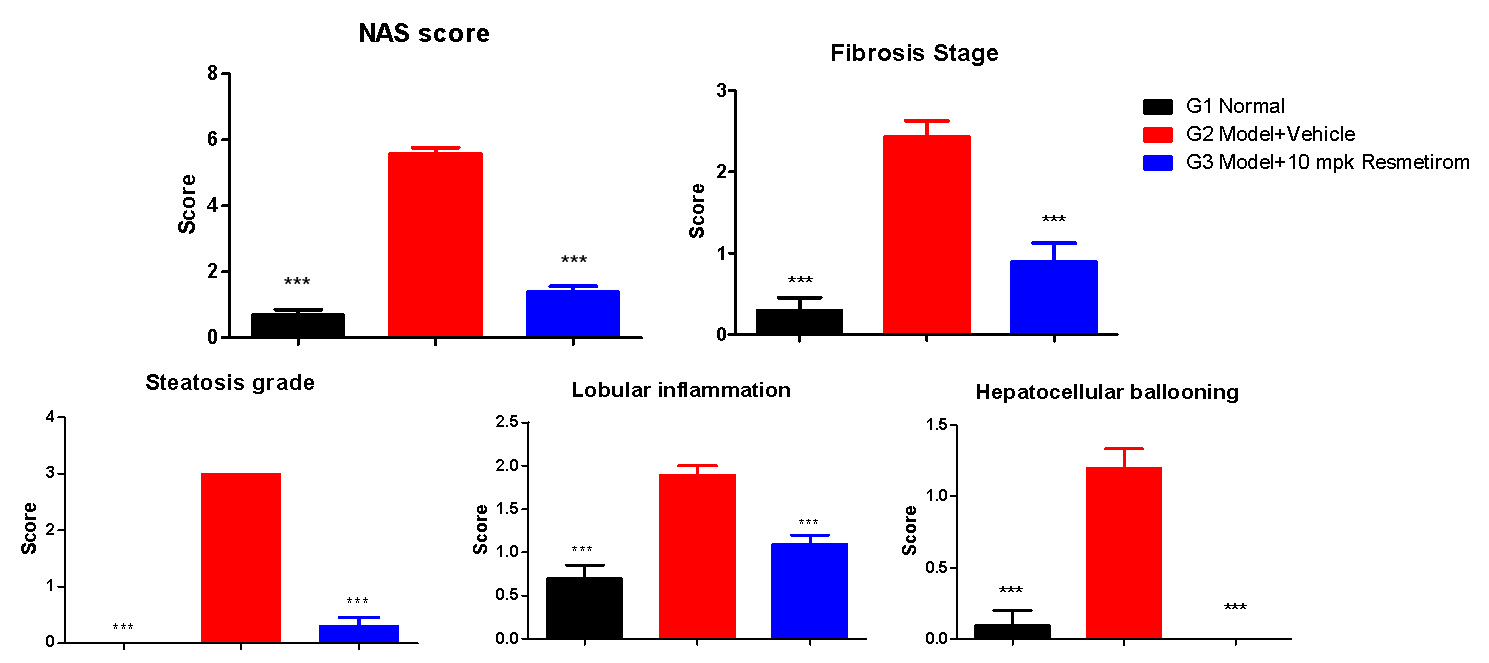
Lowered the NAS score and (modestly) fibrosis stage
OCA and Semaglutide significantly reduced steatosis, inflammation and ballooning (NAFLD Activity Score).
Modest effect on fibrosis.
Evaluation of Resmetirom in the New NASH Model
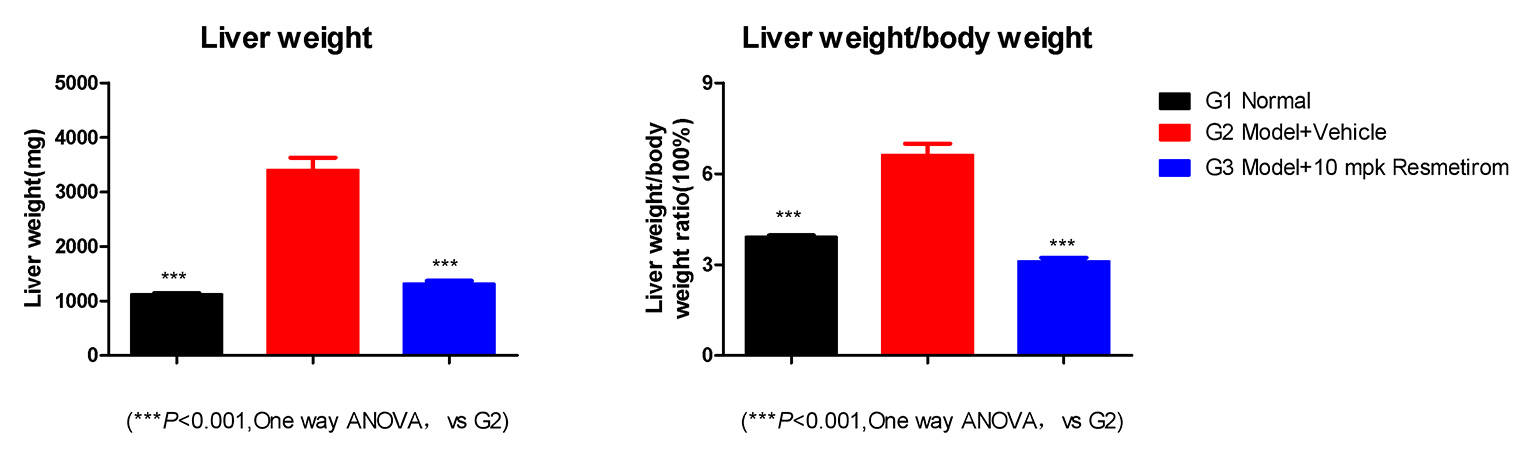
Resmetirom Significantly Lowers the Liver Weight vs the Model
Induction period: 16 weeks; treated: 8 weeks
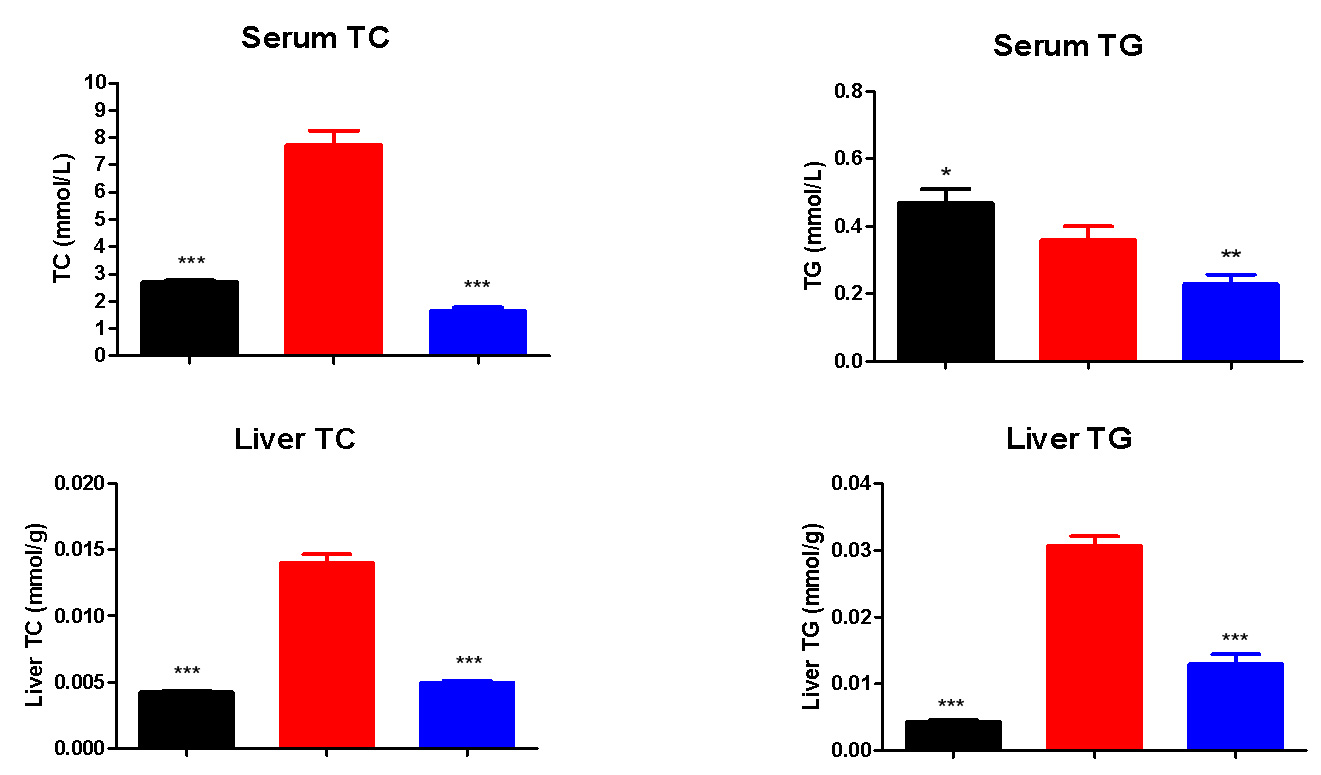
Resmetirom Significantly Lowers the Serum and Liver TC and TG vs Model
(*, P<0.05, **, P<0.01,***,p< 0.001, One way ANOVA,vs G2)
Resmetirom Significantly Improves the NAS Score and Reverses Fibrosis Stage vs Model
(*, P<0.05, **, P<0.01,***,p< 0.001, One way ANOVA,vs G2)
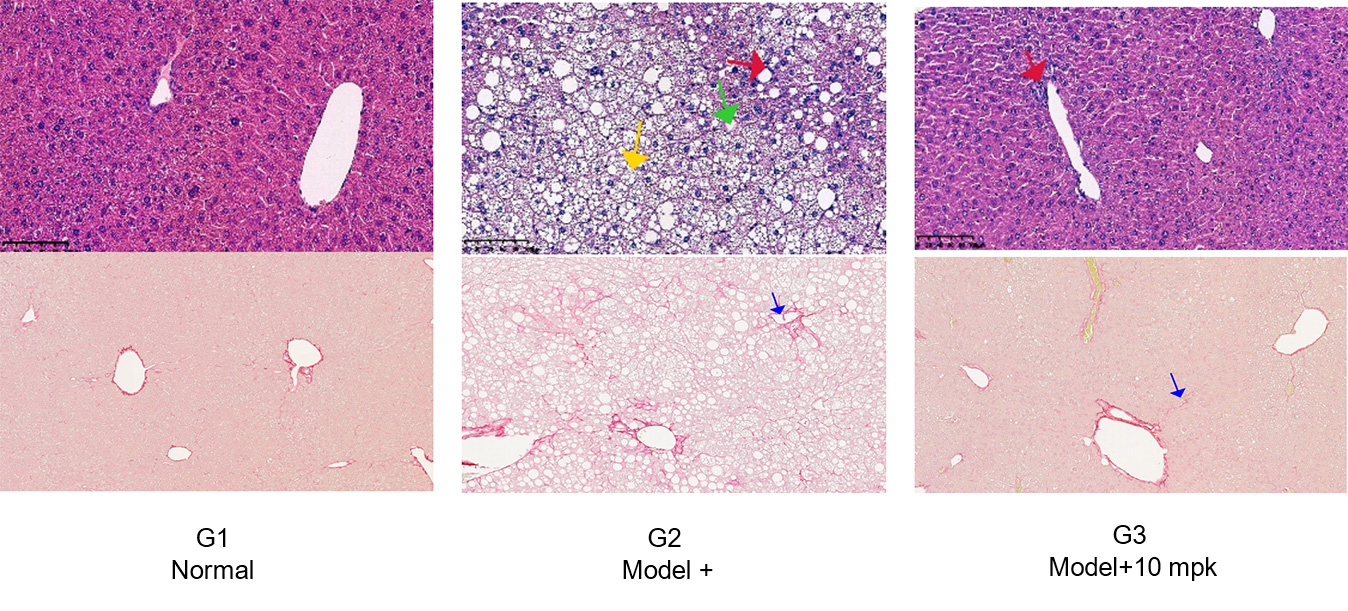
Histopathology Study Supports that Resmetirom Significantly Improves the NAS Score and Reduce Fibrosis vs
Model
Yellow arrows: steatosis
Red arrows: inflammation
Green arrows: ballooning
Blue arrows: fibrosis
HFC + Fructose Diet Induced NASH Model in Hamster
A new clinically translatable NASH Hamster model
Closely mimic the human NASH disease condition: dysmetabolic syndromes, inflammation, hepatosteatosis,
and fibrosis score
Short induction period, 8 – 12 weeks
Higher and more robust fibrosis score than the current diet-induced NASH models
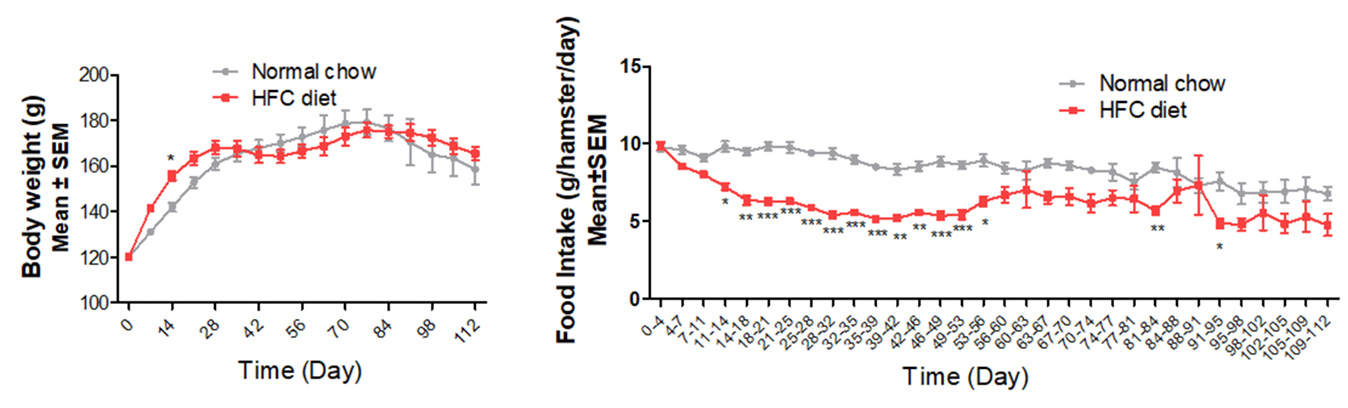
Body weight & food take of NASH Hamsters
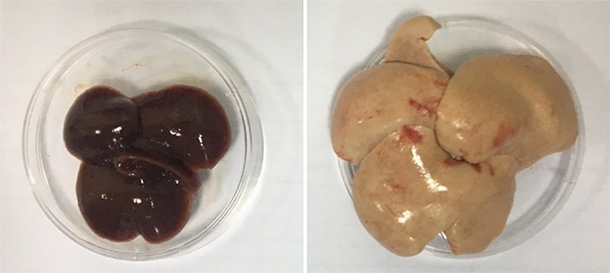
Significantly higher liver weight live to body weight ratio, ALT in HC +fructose fed Hamster
Plasma and liver lipids showed hamsters on HFC+Fructose diet developed severe dyslipidemia
Histopathological analysis showed hamsters on HFC+Fructose diet developed hallmarks of NASH
H&E staining showed steatosis, inflammation and ballooning developed in hamsters on HFC+Fructose diet.
Sirius-Red staining confirmed the fibrosis